It's never too early for breast cancer awareness!
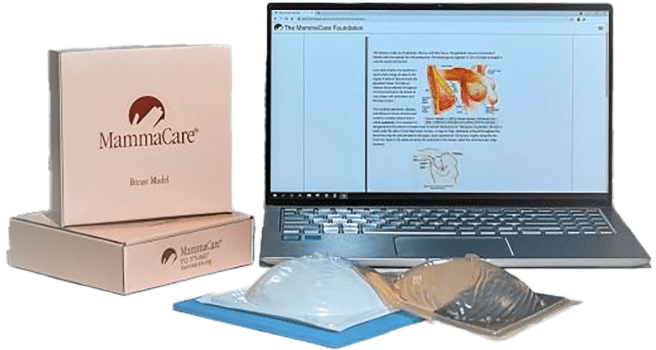
MammaCare Clinical Breast Examiner Certification
The MammaCare Clinical Breast Examination Certification course trains health care providers to perform evidence-based clinical breast exams from their own clinics via teletraining.
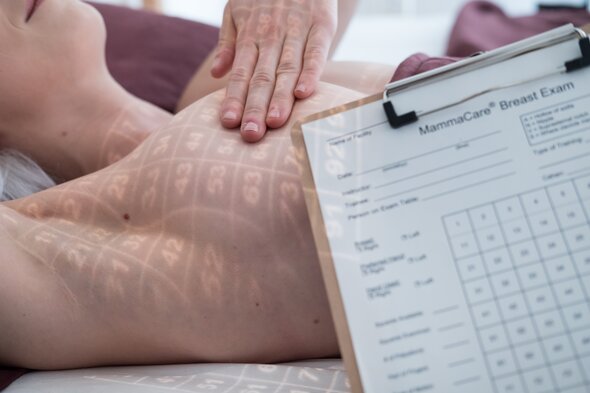
MammaCare is the recognized standard for performing clinical breast examination
Research shows that a lump is the earliest and most common sign of breast cancer. Lumps are commonly identified by palpation, an essential component of breast cancer screening that identifies interval cancers and small changes that may not be detected by imaging alone.
The MammaCare Method of breast examination is based on decades of research supported by the National Cancer Institute and National Science Foundation. Learning this method requires training the sense of touch through a series of tactually-accurate breast models and live tissue examination.
Graduates meet performance standards for sensitivity, specificity and thoroughness to earn certification as a MammaCare Clinical Breast Examiner.
No travel necessary
The MammaCare Clinical Breast Examination Certification course is delivered at your own clinic via teletraining technology.
MammaCare provides all the equipment for live, one-on-one video conference and simulation exercises. The teletraining format uses a HIPAA-compliant video feed, ensuring privacy for clinicians and surrogates during participation.
Requirements for teletraining
Trainees must provide two volunteers willing to perform as surrogate patients for a clinical breast exam to be supervised by the MammaCare instructor.
In addition, your clinic must also provide:
- IT support
- Reliable wireless internet
- Office workspace for using the Breast Exam Trainer
- A clinical exam room for the final day of training
How Clinical Breast Examiner Certification Works
Pretraining
Trainees will access the online learning platform to assess their breast health knowledge and learn to perform the 5 Ps of the MammaCare Method. Essential palpation skills are practiced using tactually-accurate breast models to identify normal breast structure and suspicious lumps.
Teletraining Day 1: Advanced CBE simulator palpation training
Trainees will prepare for advanced palpation training using the MammaCare AI Breast Exam Trainer. Trainees will achieve standards of sensitivity, specificity, and thoroughness on all four training modules before applying these skills with surrogate patients on Day 2.
Teletraining Day 2: Surrogate patient exams and final skills check-offs
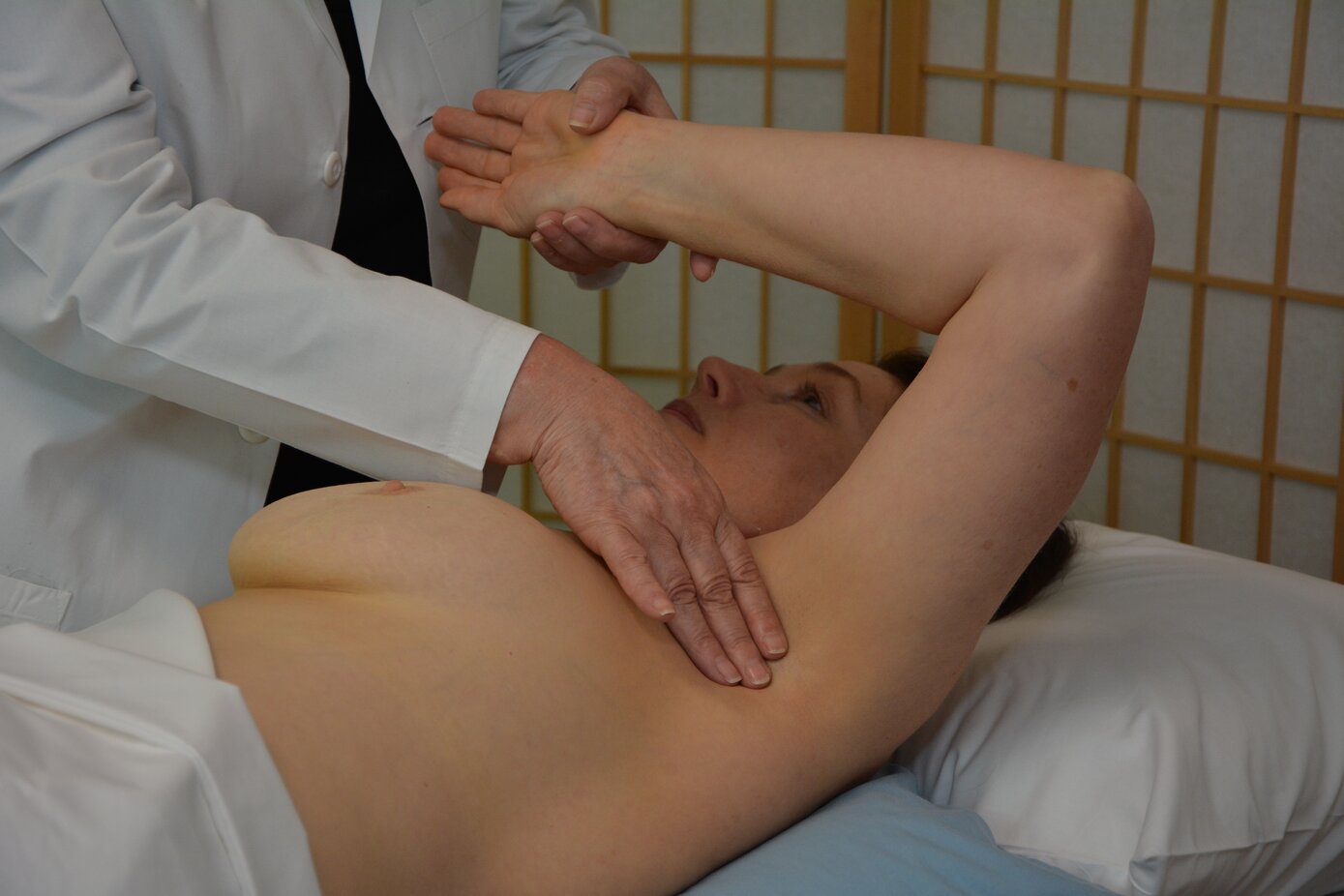
Trainees begin Day 2 by demonstrating their ability to perform the 5 Ps of the MammaCare Method on a live surrogate patient. This exam is supervised by the MammaCare instructor. During this exam, the trainee will receive feedback to improve their technique. The MammaCare instructor will perform final skill check-offs using a second surrogate patient and a series of standardized breast models to measure and document the trainee’s sensitivity, specificity, and thoroughness.
Newly certified MammaCare Clinical Breast Examiners receive:
- Certification credentials: CBE-MC
- Certificate of Completion from The MammaCare Foundation (valid for 3 years)
- 9 continuing education credits approved by the Florida Board of Nursing (if applicable)
- MammaCare Clinical Breast Examiner Pin
- Two standard lump displays for patient education
- MammaCare Professional Learning System (MLS-1), which includes two tactually-accurate breast models (to keep)
- Skill maintenance breast model for post-training practice (to keep)
“The increased ease of accessing the materials online and the trainer were invaluable to me and my team. Taking time away from patient care is basically eliminated with this method.”
– Graduate from Mount Desert Island Hospital
“The instructor was just SPECTACULAR!!! She made me feel at ease and confident. The course was so well organized with plenty of time to learn properly. I honestly loved everything about it!!”
– Graduate from Lehigh Valley Hospital
“What I liked best about the course was the learning environment and the adaptability of the course offering (teletraining works well).”
– Graduate from Northwest Florida Community Hospital
“A huge thank you to everyone that had a part in my MammaCare experience. I cannot wait to help my patients and ensure they are receiving the highest quality breast care available. THANK YOU!”
– Graduate from Northwest Florida Community Hospital
“I now have more confidence in my examination skills and felt the simulator was very helpful for providing feedback!”
– Graduate from Mercy Hospital St. Louis
I am interested in becoming certified as a Clinical Breast Examiner

The MammaCare® Foundation is a 501(c)(3) non-profit dedicated to training every hand that examines a woman, including her own. The MammaCare Method is the recognized standard for performing and teaching clinical and personal breast examination. MammaCare provides clinical certification services and high-fidelity simulation-based training.
Phone
+1 352 375 0607
Address
930 NW 8th Ave,
Gainesville, FL, 32601
United States

